Clinical Exchange Document
Description
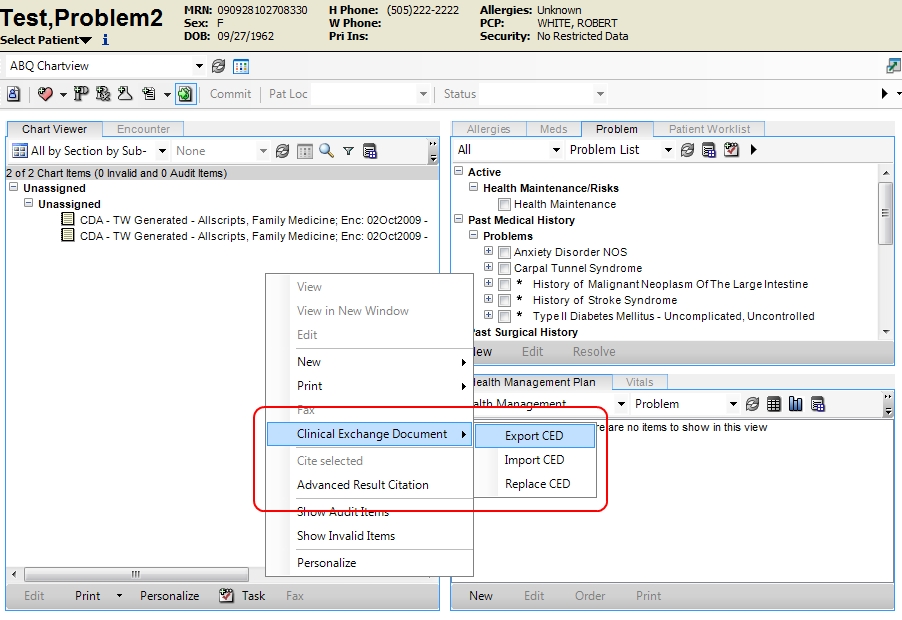
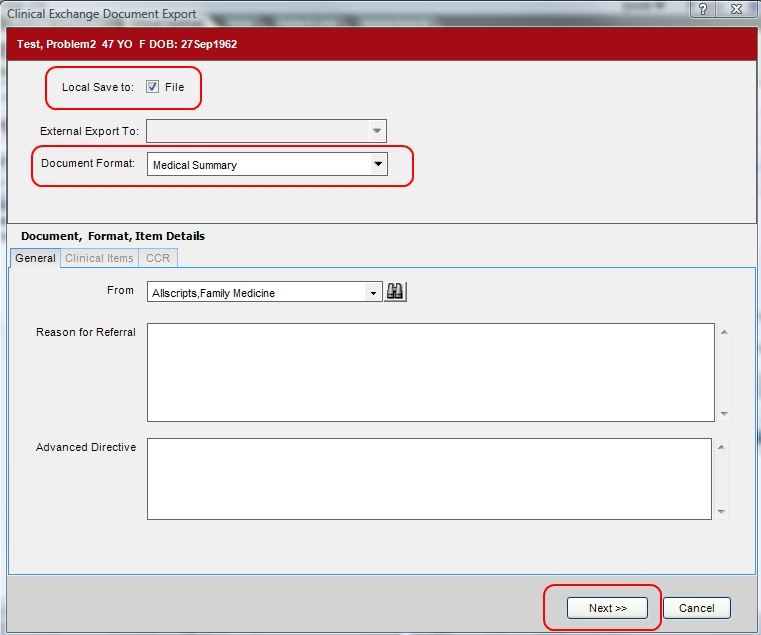
A Clinical Exchange Document or CED is a generally accepted format of patient data that is used to share information between systems. CCR and Medical Summary (a CDA) are the two CED formats that are supported in V11. CED is an Allscripts term to cover all the different formats that are supported. Both are xml documents, and both are essentially encounter summaries containing allergies, medications, immunizations, problems, vitals, results.
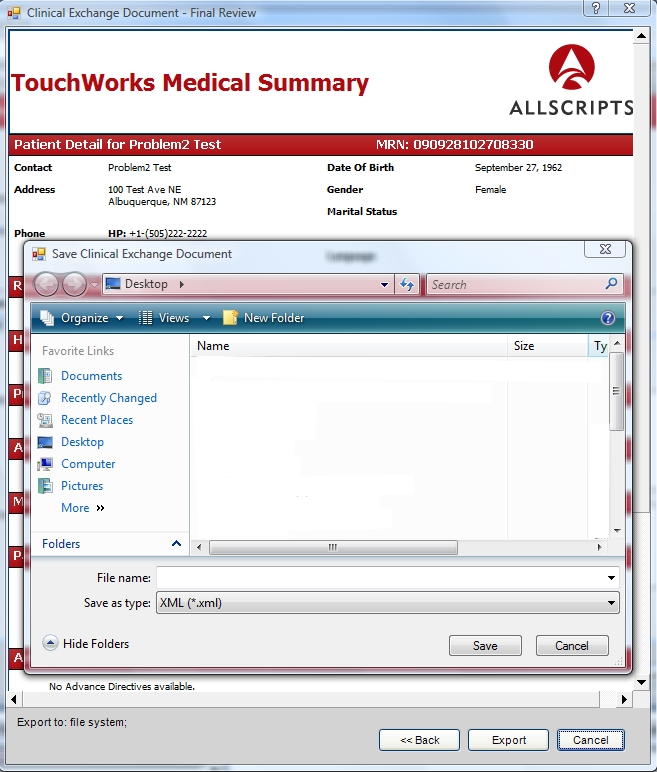
These extracts can be printed or saved as an xml document. The xml document can be imported into other TouchWorks systems or other EHRs that support the format.
CCD Documents
CCD documents combine the benefits of the CCR with the CDA specifications.
From the HL7 CCD Handbook - The HL7 Clinical Document Architecture (CDA) is an XML-based markup standard intended to specify the encoding, structure and semantics of clinical documents for exchange. CDA is part of the HL7 version 3 standard. CDA documents are persistent in nature. The CDA specifies that the content of the document consists of a mandatory textual part (which ensures human interpretation of the document contents) and optional structured parts (for software processing). The structured part relies on coding systems (such as from SNOMED and LOINC) to represent concepts.
From Wikipedia - CCD was developed as a collaborative effort between ASTM and HL7, combining the benefits of ASTMs Continuity of Care Record or CCR and the HL7 Clinical Document Architecture (CDA) specifications. It is intended as an alternate implementation to the one specified in ASTM ADJE2369 for those institutions or organizations committed to implementation of the HL7 Clinical Document Architecture.
Allscripts & CCD
TouchWorks v10.1 HealthMatics 2006
- Began to expose the Export and Import of one document format CCR, but using a preliminary schema. Allscripts was ready to try this in product before the ASTM had a ballot for an official version of CCR.
TouchWorks v10.2.3 and HealthMatics 2006
- Exports only CCR V1.0, the official version, but continues to import either the preliminary as well as the official version.
TWEHR v11 and HealthMatics 2007
- Supports multiple document formats:
- CCR preliminary schema
- CCR V1.0
- CDA/CRS, better known as Medical Summary, the HL7 specified format for referrals and encounter summary
TWEHR v11.1.7
Microsoft HealthVault Integration
- HealthVault-EHR relationship addresses strong demand for clinical data sharing between portals and EHR
- Touchworks EHR links to patient’s HealthVault account
- Bi-directional data sharing
- Patient’s clinical information can be pulled from patient’s HealthVault account via a Clinical Content Document (CCD) into TWEHR
- Data elements enter TWEHR as discrete and must be verified by the practice before being labeled as “active”
- TWEHR can push the patient’s clinical data to HealthVault
- Benefits
- Save practice time creating/updating patient’s chart
- Partner with Microsoft, a consumer health platform leader
- Provide access for patients to be involved in the management of their own care
Duplicate Checking/Trusted Source
- Clinical Exchange Documents received in the CCD format:
- Clinical Exchange Documents that already exist in the system will not import to prevent duplicate information
- When discrete items (Problems, Medications, and Allergies) are imported from a Clinical Exchange Document and might be a duplicate, they are given the status of “Potential Duplicate”
- When Clinical Exchange Documents are received from a source that is considered “Trusted”, Touchworks EHR will attempt to add the Problems, Medications, and Allergies with a “Verified” status
- If a Clinical Exchange Document, received from a particular source should be managed differently than the default, it is managed in the Clinical Document Target dictionary
- Clinical Exchange Documents received in any format:
- The preference CreateQuarantinedItemsFromImportedCEDs is removed and default settings are managed using the Unidentified Source enforced item in the Clinical Document Target dictionary